March 14, 2025
Authors: André Nimtz & Maren Fichtner
Reading time: approx. 20 minutes
Definition: What is quality control?
When we talk about quality in production, we come across a variety of terms. They range from quality management and quality assurance to quality testing and quality control. They are often used interchangeably, but the crux of the matter is that they all have their own meaning and relevance.
In this article, we will focus on quality control and its actual meaning. Quality control refers to procedures and processes that are used to check a product or service against predefined quality criteria. Such requirements can be, for example, specific production specifications, certain standards or customer requirements.
It is important to note that quality control has a clear product-related focus. It is specifically about checking a (partially) manufactured product - with the aim of identifying errors, defects or deviations.
Incidentally, quality control is also often referred to as quality inspection during research. However, we are talking here about quality control as an internal production process within quality assurance, whereas a quality inspection can also be carried out by external service providers.
Quality control vs. quality assurance
Quality control and quality assurance - aren't they actually the same thing? Not at all. Both concepts are part of quality management and represent different levels of it.
Quality assurance is a process-related concept. Roughly speaking, it deals with the advance planning of inspection concepts as well as inspection and measurement systems along the entire value chain.
Quality control, on the other hand, is product-related. It is specifically concerned with checking whether the manufactured product meets the defined quality criteria.
You can read more about the differences between quality control and quality assurance in our blog post on quality assurance in production.
The tasks of quality control all revolve around – you guessed it – ensuring product quality in accordance with predefined specifications. This includes:
![]()
Preparation of the test
All necessary test materials, equipment and test plans are provided.
![]()
Carrying out the test
On the one hand, this includes the actual inspection, for example using reference samples or inspection plans. On the other hand, quality control ensures that all relevant checks have been carried out.
![]()
Approval of products
Does the product or service meet the specifications? Then it is approved by quality control. If not, approval is not granted and follow-up measures must be initiated.
![]()
Documentation
Finally, the results must be documented for internal and external use. Quality control also ensures that tested products are correctly labeled - for example as “rework”, “released” or “reject”.
Does quality control have to be independent of production?
It is not uncommon to find statements to the effect that quality control can only function properly if it is independent of production. But is that really true?
Let's say so: Yes and no. As is so often the case, it depends on a number of conditions. One condition can be the industry. In highly regulated sectors such as the pharmaceutical industry or automotive engineering, the situation is quite different from that in mechanical engineering. Here, quality control must be carried out by specialists.
In manufacturing and assembly, quality control can largely be delegated to the workers themselves. The key to this is a system that enables them to carry out appropriate checks and reliably record the results. A worker guidance system provides valuable services here.
In a nutshell: You can either check randomly or check everything. What are the differences?
Sampling
Sampling is particularly suitable for mass production. With random sampling, you check the quality either randomly or according to a predefined number of products or shots. You define in advance how large the number of products to be inspected is and how many defects or faults per sample are acceptable.
One hundred percent testing
If you manufacture in particularly small batch sizes or customized products, it makes sense to test every product (or service). Of course, you must also define criteria in advance according to which the products are tested. The only difference is that you check every product and may invest more time in an in-depth inspection.
The intelligent hybrid form: dynamic test steps
You don't have the capacity to carry out a large number of product tests? But you also don't want to rely on static testing every X shots? How about dynamic inspection steps?
Our worker guidance system weasl can - in cooperation with your peripheral systems - dynamically intersperse test steps according to defined criteria.
If decisive quality features deviate from your targets, the system can display additional inspection steps to your workers. This allows you to carry out quality checks dynamically, depending on the situation and as required.
Beyond this, you can also implement decision trees directly in the editor in weasl, which trigger inspection or rework steps as soon as a certain number of defects have been recorded, for example.
Targeted quality controls pay off for your company on many levels. And these range from the optimization of your own processes and procedures to your competitiveness.
Your good market image
High quality standards strengthen your reputation with your customers. High quality at a fair price pays off much more for you and your customers than questionable quality at a knockdown price.
Convinced customers who return
If you can ensure that all the quality standards required by the customer have been met, you are not just selling a product. You gain a customer who will come back and recommend you to others.
Securing your quality level
Once the right measures for effective quality control have been established, you can offer products and services with a consistently high level of quality.
Fewer errors, more profitability
Quality controls help to detect errors at an early stage and prevent defects. You always deliver flawless goods and thus avoid economic and possibly personal damage. Fewer defects also result in fewer returns and reworking, which in turn increases your profitability.
Promoting improvement processes
Continuous quality controls allow you to identify and eliminate weaknesses in your processes time and time again. This promotes the continuous improvement process in your company.
Compliance with legal regulations
Do you have to comply with legal regulations or certain standards? Then use quality controls at the relevant points to ensure and document compliance.
More sustainability
Quality controls can contribute to greater sustainability in various ways: fewer rejects means less waste of resources. Fewer complaints means fewer transportation routes.
There is actually no real argument against sensibly established quality controls. On the contrary: there are a few more arguments that you should keep in mind when deciding on an effective quality control system:
- If complaints arise as a result of inadequate quality control, you will have to spend a lot of time processing them - combined with costs that cannot be ignored.
- In industry in particular, you should bear in mind that if you have to rectify quality problems on site at the customer's premises, you will be faced with high travel costs and further loss of time. Or you may have to establish additional, expensive technologies for remote services.
- Recurring quality deficiencies can sooner or later lead to the loss of your customers. Negative reviews or word of mouth will do the rest.
- If compliance with certain standards and regulations fails due to poorly established quality controls, there is also the risk of losing a license or approval in your industry.
Of course, quality controls can initially incur costs and require technological adjustments. However, the benefits you derive from established testing processes are significantly greater.
Quality controls can be found at various points in the production process - from incoming goods to outgoing goods. Let's take a closer look at the four basic checks.
Incoming Quality Control (IQC)
As the name suggests, a quality check is carried out on the incoming product or component when the goods are received. The aim is to ensure that the outgoing product complies with the specifications and that there are no errors at this stage that may no longer be traceable later.
In-Process Quality Control (IPQC)
In-process Quality Control (IPQC) involves continuous quality checks during production - i.e. everything that occurs between the incoming product and the end product. IPQC is particularly relevant in the case of strict regulations or requirements.
Final Quality Control (FQC)
With Final Quality Control (FQC), we have arrived at the last inspection and the final tests of the product before delivery. The aim here is to ensure that the necessary standards for the customer or the market have been met.
Outgoing Quality Control (OQC)
After the final inspection, the product is packaged and provided with the necessary documents. Outgoing Quality Control (OQC) checks that the packaging is free of defects and that all relevant documents are enclosed.
All industries can benefit from quality control. However, there are some industries that are particularly reliant on quality control. In principle, this applies to all industries that manufacture products or offer services that pose a potential risk of harm to humans. These include, for example
- the automotive industry
- the pharmaceutical industry
- medical technology
- the construction industry
- manufacturers of cosmetics
- food producers
- animal feed manufacturers
- etc.
For companies in the medical and food sectors in particular, guidelines have already been established that specify detailed quality standards. These include, for example:
- GxP or “Good Practice” as a cross-industry quality guideline
- GmP or “Good Manufacturing Practice”: “Good Manufacturing Practice” comprises guidelines for the quality assurance of production processes and environments that are used in the manufacture of pharmaceuticals, active ingredients, cosmetics, food and animal feed.
In order to optimally comply with these respective standards, companies need reliable quality assurance processes and - in practice - the corresponding quality controls.
Step 1
Define quality standards
So that the inspector knows what is to be inspected and how, the quality standards must first be defined as a quality assurance measure. To do this, quality assurance must
- define measurable and evaluable quality criteria
- define corrective measures in the event of deviations
- record and formalize specifications, for example through standards or customer requirements
- provide references, test plans or reference samples
Step 2
Production monitoring through regular quality controls
The next step involves continuous monitoring of the production processes from a quality perspective. For example, you can have visual inspections carried out by the worker using sensors or cameras. Quantitative tests and (performance) measurements - for example electrical tests, chemical analyses or stress tests - are also an effective means of quality control.
The aim of quality control is to identify deviations and errors by comparing the recorded results with the specified quality standards. If errors occur, you initiate appropriately defined measures to rectify them (rework, training, etc.).
Does everything fit? Then quality control releases the product as a good part.
Step 3
Documentation
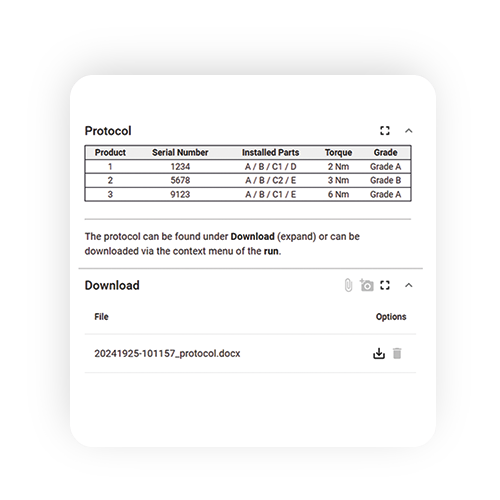
Of course, quality control also includes recording the results. Among other things, the following are recorded in such a protocol
- the name of the material or product
- the batch designation, manufacturer and/or supplier if applicable
- information on specifications and test procedures
- information on the test equipment used
- the test results (incl. observations, calculations, certificates of analysis)
- the date and location of the test performed
- the names of the persons carrying out the test
- the names of the verifying persons, if applicable
- the result in the form of a clear decision on approval or rejection
Step 4
Supporting continuous improvement processes
In this step, you move from quality control to quality improvement and use the findings from the audits to identify possible trends and promote continuous improvement processes. This in turn has a positive impact on your quality standards.
CAQ or QM software
There is a suitable solution for everything, including quality management. Computer-aided quality assurance (CAQ) or quality management software makes it easier to record and evaluate quality-related data. This data can in turn be used to derive conclusions for the further planning of quality assurance and quality control measures.
Worker guidance systems
With a worker guidance system, you can guide employees digitally through inspection steps or complete inspection plans. As part of the digital work instruction, you specify standardized quality data and record collected or entered data directly in the system.
Internet of Things (IoT)
The Internet of Things describes the networking of devices and systems - including in production. Networked sensors, cameras or control systems allow you to record data in real time and detect and rectify anomalies or quality deviations more quickly.
Artificial Intelligence (AI) und Machine Learning
Intelligent algorithms enable the centralized or decentralized evaluation of large data sets. They can predict possible errors or system failures that could affect the final quality of the product.
Yes, we've already talked about the benefits of quality control above. But we have another bonus for you: digitized quality control offers further advantages:
Increased efficiency
Digital systems document automatically, replace manual input and make input errors or subjective interpretations of errors obsolete.
Accessibility
If you map quality controls with a worker guidance system, for example, your employees have access to all required and relevant documents and information at all times.
Traceability
Data and information are recorded digitally directly in the process and are therefore immediately available and traceable. This enables seamless traceability.
Integration of peripheral systems
Digitization allows you to achieve consistency in your systems. You can easily exchange digitally recorded quality data across departments or along the supply chain.
Data security and compliance
Digital quality documentation can be digitally secured in a watertight manner – including access controls, encryption and back-ups.
Predictive Quality and CIP
The digital availability of data and its analyzability enable you to make predictions and continuously improve the quality of your processes and products.
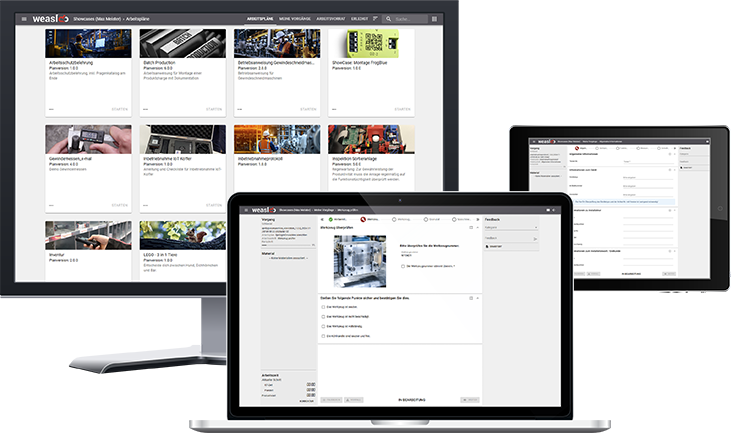
For effective quality control, it is best to use a digital system - especially if you want to hand over inspection steps to your employees in production and assembly. Our worker guidance system weasl provides the perfect basis for this.
Test plans as work instructions
With weasl, you can implement inspection plans as easy-to-understand, multimedia-supported step-by-step work instructions. You can use a role-rights system to define which inspections can be carried out by a worker and which can only be carried out by trained inspection personnel. In this way, you empower your workers to carry out worker self-inspection and strengthen their quality awareness.
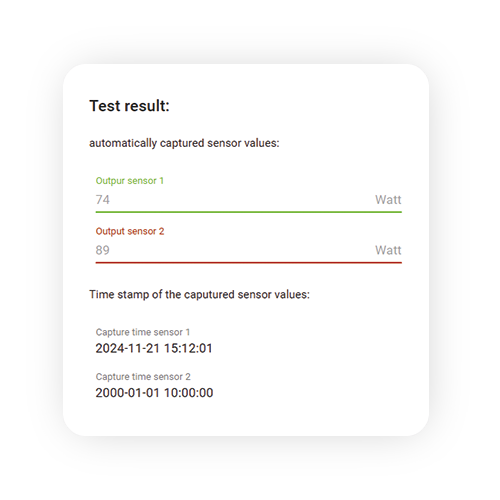
Securely record quality data
You can easily validate quality-relevant entries with weasl by marking the corresponding input fields with color cues, for example. If an input value exceeds the tolerance range, the worker is informed by a red message text, for example. You can also prevent incorrect entries through standardization - for example, by only allowing measurements in defined numerical ranges and units.
Creating consistency
weasl communicates with your systems, tools and test equipment. If the required inspection tools are connected to weasl, your workers can automatically read out inspection data and transfer it to the system. Communication - for example in the event of queries or support requests - also takes place directly in the system thanks to the feedback function.
Document quality controls digitally
With weasl, you record and document all work steps, results and input values automatically. This prevents media breaks and the resulting errors and loss of time. At the end of a work or test plan, weasl automatically generates a log with all the data and information.
Conclusion
Without effectively established quality controls, any production process will struggle in the long term. Customers expect quality, lean production processes require error-free work and, last but not least, quality defects are also a cost driver.
Quality controls do not necessarily have to be a separate, extensive complex in your production. Simple but highly efficient measures can already be implemented as part of worker self-checks. With digital support such as a worker guidance system, you can make work easier for yourself and your employees by integrating quality inspections directly into your processes, recording data directly in digital form and ensuring automatic verification and documentation.
Implement quality control easily - with weasl
Enable your workers to carry out quality checks themselves in the production and assembly process. Record quality data digitally and establish effective quality assurance processes - with our weasl worker guidance system.
Experience for yourself what weasl can do - in our free showcase environment.